 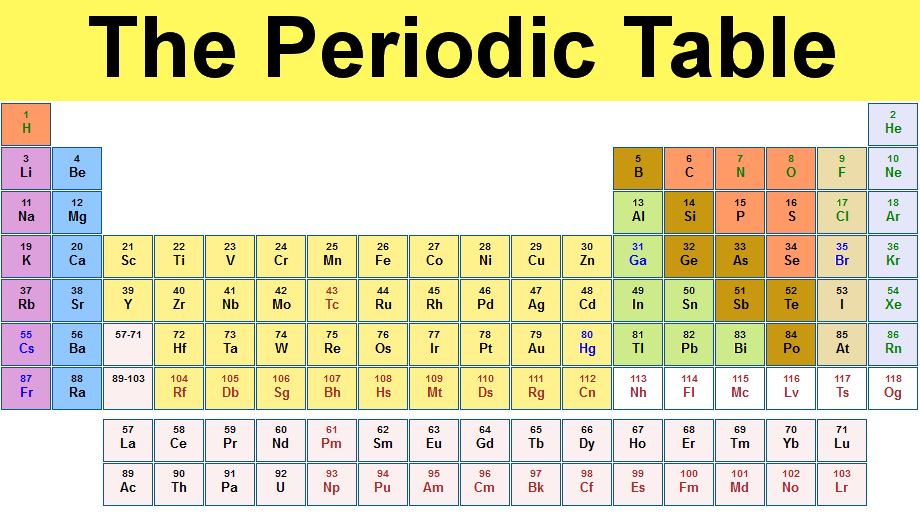
In Chapter 1, we described Dalton’s theory that each chemical compound has a particular combination of atoms and that the ratios of the numbers of atoms of the elements present are usually small whole numbers. However, as you can see on the periodic table, sulfur is listed as 32.07 amunot 31.97 This is because the periodic table lists atomic weightsthe averages of the atomic mass for each one of the element’s stable isotopes. 
This structure contains sulfide ions on the lattice points of an FCC lattice. (8 atoms)(15.9994 amu/atom) = 127.9952 amuįormula mass of Ca 3(PO 4) = 310.177 2amu Going back to 32 S, we know that because its molar mass is 31.97 grams per mole, its atomic mass must be 31.97 amu. The cubic form of zinc sulfide, zinc blende, also crystallizes in an FCC unit cell, as illustrated in Figure 10.61. Atomic weight of Lithium (Li): 6.941 (Ref: jlab-ele003) Atomic weight of chlorine (Cl) : 35.4527 (Ref: Pubchem-Chlorine, Jlab-ele003) Step 3: Calculate the molecular weight of Lithium chloride by adding the total weight of all atoms. (2 atoms)(30.973761 amu/atom) = 6.0474 amu Step 2: Find out the atomic weights of each atom (from the periodic table). 48 grams O×1 mole O/15.999 grams O3.0002 moles O Second, we can see that there are 2 oxygen atoms in the molecule of CO2. Under standard conditions, it is the least. Formula: LiCl Hill system formula: Cl1Li CAS registry number: Formula weight: 42.394 Class: chloride Colour: white Appearance: crystalline. It is a soft, silvery-white alkali metal. Molar mass of K 39.1 g Molar mass of Mn 54.9 g Molar mass of O 16.0 g Molar mass of KMnO 4 39.1 g + 54.9 g + (16.0 g x 4) Molar mass of KMnO 4 158.0 g Use this number to convert grams to moles. 1.5 moles To calculate the number of moles of CO2 in a sample that contains 48 grams oxygen, we need to convert the grams of oxygen into moles of oxygen using our molar mass from the periodic table. Lithium is a chemical element it has symbol Li and atomic number 3. \right ) \right ]=310.177\ amu \notag \) Atoms To convert grams to moles, the molar mass of the solute is needed, which can be found on certain periodic tables. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
